Introduction: Novel T-cell redirecting therapies (TCR), including chimeric antigen receptor T-cell therapy (CAR-T) and bispecific antibodies (BsAbs), are important new treatment options for patients (pts) with relapsed/refractory multiple myeloma (RRMM) but result in a new unmet need for pts who relapse following these therapies. Talquetamab is the most advanced BsAb therapy targeting G protein-coupled receptor family C group 5 member D (GPRC5D), a novel antigen that is overexpressed on malignant plasma cells but is expressed at low levels on B cells and bone marrow progenitors. Previous results from the phase 1/2 MonumenTAL-1 study (NCT03399799/NCT04634552) demonstrated deep and durable responses with talquetamab in pts with RRMM, with an overall response rate (ORR) of >71% in 288 pts naive to TCR and 65% in 51 pts with prior TCR. Here, we present updated results in pts with prior TCR, including an additional 19 pts enrolled since the prior analysis.
Methods: Eligible pts for phase 1 of the study had progressed on or could not tolerate established multiple myeloma therapies.In phase 2, eligible pts had been exposed to ≥3 prior lines of therapy including a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 antibody. The current analysis includes all pts across phase 1 and 2 of the study who received 1 of the 2 recommended phase 2 doses of subcutaneous talquetamab (0.4 mg/kg weekly [QW] or 0.8 mg/kg every other week [Q2W]) and who were exposed to a TCR (prior CAR-T and BsAb), as well as pts exposed to both a TCR and a B-cell maturation antigen (BCMA)-targeting antibody-drug conjugate (ADC).
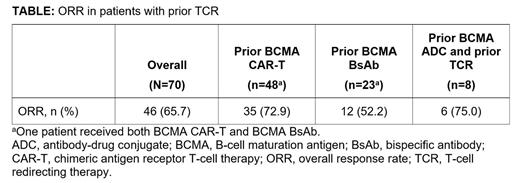
Results: As of Jan 17, 2023, 70 pts were enrolled with prior TCR. Overall, 44 (63%) were <65 years old; 65 (93%) received ≥4 prior lines of therapy; 58 (83%) and 29 (41%) were triple-class and penta-drug refractory, respectively; 21 (30%) had extramedullary disease, 24 (34%) had high-risk cytogenetics, and 12 (17%) had International Staging System stage III disease. Fifty of 70 pts received prior CAR-T (48/50 anti-BCMA CAR-T), 25 pts received prior BsAb (23/25 anti-BCMA BsAb), and 5 pts received both. Eight of 70 pts received prior treatment with BCMA ADC. Among BCMA-exposed pts, ORR was comparable between pts receiving prior BCMA CAR-T and prior BCMA ADC, and similar to the overall population; ORR was lower in pts receiving prior BCMA BsAb (Table). Median duration of response (mDOR) was similar between the overall population (12.3 mo; 95% CI, 6.5-not estimable [NE]) and pts with prior BCMA CAR-T (12.3 mo; 95% CI, 4.8-NE), but was shorter in pts with prior BCMA BsAb (6.5 mo; 95% CI, 1.9-NE). In the prior CAR-T group, ORR was comparable in those who received CAR-T as immediate prior line vs at any prior line of therapy before talquetamab (75.9% [22/29] vs 71.4% [15/21], respectively). In the prior BsAb group, ORR was lower in those who received BsAb as immediate prior line vs at any prior line of therapy before talquetamab (28.6% [2/7] vs 61.1% [11/18], respectively). In the prior BsAb group, ORR was 62.5% (5/8) in pts who had ≥9 mo interval between last dose of the prior BsAb and talquetamab, 50% (3/6) in those who had ≥6 to <9 mo interval, and 45.5% (5/11) in those who had <6 mo interval.
Conclusions: These updated results from pts with prior TCR in the phase 1/2 MonumenTAL-1 study show continued strong efficacy of GPRC5D-targeting talquetamab across populations exposed to TCR (predominantly anti-BCMA), with notable ORR of 73% and mDOR of >1 year in the post-CAR-T setting. More than 50% of pts exposed to prior BsAb responded, and outcomes data in these pts may offer insight into optimal sequencing. Together with previously published data, these results support talquetamab as a versatile treatment option that provides robust responses in TCR/BCMA-naive and TCR/BCMA-exposed pts with RRMM.
Acknowledgments: This study was funded by Janssen Research & Development, LLC. Medical writing support was provided by Michelle Yang, PharmD, of Eloquent Scientific Solutions, and funded by Janssen Global Services, LLC.
Disclosures
Jakubowiak:BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi-Aventi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Karlin:AbbVie, Amgen, Celgene, Janssen, Sanofi, Takeda: Honoraria; Amgen, Celgene, GSK, Janssen, Takeda: Consultancy. Chari:Janssen: Consultancy, Other: Advisory Board, Research Funding; AbbVie: Other: Advisory Board; Genentech: Other: Advisory Board; Secura Bio: Consultancy, Other: Advisory Board; Shattuck Labs: Other: Advisory Board; Antengene: Consultancy; Millenium/Takeda: Consultancy, Research Funding; Seattle Genetics: Other: Advisory Board, Research Funding; Sanofi: Other: Advisory Board; Karyopharm: Other: Advisory Board; Amgen: Consultancy, Other: Advisory Board, Research Funding; BMS: Consultancy, Other: Advisory Board, Research Funding; Glaxo Smith Kline: Other: Advisory Board. Rasche:Pfizer: Consultancy, Honoraria; Amgen: Consultancy; BMS: Consultancy, Honoraria, Research Funding; GSK: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Skyline Dx: Research Funding; Sanofi: Consultancy, Honoraria; Roche: Honoraria. San-Miguel:Abbvie: Consultancy, Other: Advisory Board; Amgen: Consultancy, Other: Advisory Board; BMS: Other: Advisory Board; Celgene: Other: Advisory Board; GSK: Other: Advisory Board; Haemalogix: Other: Advisory Board; Janssen-Cilag: Other: Advisory Board; Karyopharm: Other: Advisory Board; MSD: Other: Advisory Board; Novartis: Other; Takeda: Other: Advisory Board; Regeneron: Other: Advisory Board; Roche: Other: Advisory Board; Sanofi: Other: Advisory Board; SecuraBio: Other: Advisory Board. Campagna:Janssen: Current Employment, Current equity holder in publicly-traded company, Current holder of stock options in a privately-held company. Hilder:Janssen: Current Employment, Current equity holder in private company. Masterson:Janssen R&D: Current Employment, Current equity holder in private company. Qin:Janssen: Current Employment. Renaud:Johnson & Johnson: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Tolbert:Janssen: Current Employment. Vishwamitra:Johnson & Johnson: Current Employment, Current holder of stock options in a privately-held company. Skerget:Janssen: Current Employment, Current equity holder in publicly-traded company. Moreau:janssen, celgene BMS, abbvie, sanofi, amgen, takeda, pfizer: Honoraria, Other: advisory boards; GSK: Honoraria, Other: Advisory Board.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal